Dual GIP/GLP-1 Receptor Agonists Market: Redefining Obesity Treatment Through Incretin-Based Innovation
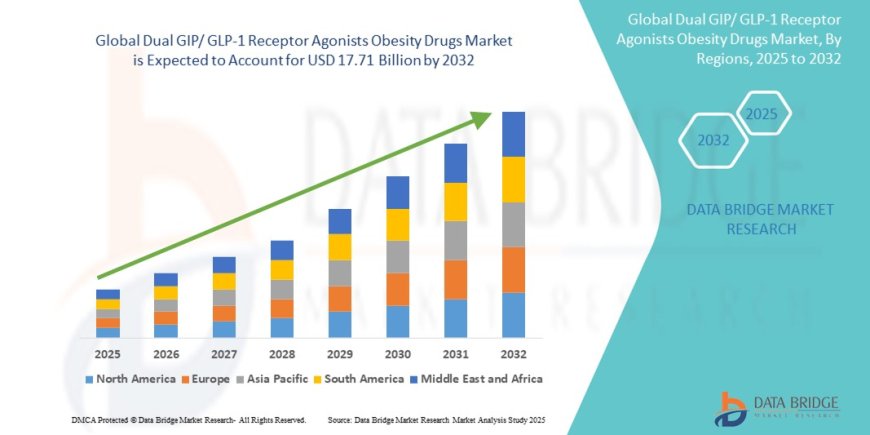
The global dual GIP/ GLP-1 receptor agonists obesity drugs market size was valued at USD 4.75 billion in 2024 and is expected to reach USD 17.71 billion by 2032, at a CAGR of 17.85% during the forecast period
Introduction
Dual GIP/GLP-1 receptor agonists represent a new class of anti-obesity drugs that simultaneously activate glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptors. This dual mechanism enhances glycemic control, suppresses appetite, and promotes significant weight loss. As obesity rates surge globally, these therapies are reshaping the treatment paradigm with superior efficacy compared to monotherapies.
Market Overview
The global market was valued at USD 4.75 billion in 2024 and is projected to reach USD 17.71 billion by 2032, growing at a CAGR of 17.85%.
-
North America leads with 45.6% share, driven by high obesity prevalence, advanced healthcare infrastructure, and early adoption of drugs like Tirzepatide (Mounjaro)
-
Asia-Pacific is the fastest-growing region, fueled by urbanization, rising disposable income, and increasing obesity rates in countries like India, China, and Japan.
-
Europe shows steady growth due to regulatory support and expanding access to GLP-1 therapies
Market Segmentation
By Drug Type
-
Tirzepatide-based (dominant, ~47.8% share in 2024)
-
Semaglutide-based
-
Others (e.g., Retatrutide, PN-477, GZR18)
By Route of Administration
-
Subcutaneous
-
Oral (emerging segment with candidates like PN-477o)
-
Others
By Indication
-
Obesity
-
Type 2 Diabetes
-
Cardiovascular Disorders
-
Others
By Distribution Channel
-
Hospital Pharmacies
-
Retail Pharmacies
-
Online Pharmacies
By End User
-
Hospitals
-
Specialty Clinics
-
Homecare Settings
Key Trends
-
Superior Weight Loss Outcomes: Tirzepatide and emerging triple agonists (GLP-1/GIP/GCG) show >20% weight reduction in trials.
-
Oral Formulations: Protagonists PN-477 aims to offer once-daily oral dosing with triple receptor activation
-
Pipeline Expansion: Drugs like Retatrutide and Survodutide are advancing multi-pathway approaches for metabolic health
-
Rapid Market Penetration: Mounjaro captured 8% of Indias obesity drug market within 3 months of launch
-
Combination Therapies: Dual and triple agonists are being explored for broader comorbidity management including cardiovascular and liver health
Challenges
-
High Cost and Accessibility: Injectable therapies remain expensive, limiting access in low-income regions
-
Regulatory Complexity: Diverse approval pathways across geographies slow global rollout
-
Adverse Effects: GI side effects and long-term safety require ongoing monitoring
-
Patent Expiry and Generic Competition: Semaglutide generics expected post-2026 may disrupt pricing dynamics
Growth Drivers
-
Global Obesity Epidemic: Over 650 million adults classified as obese globally
-
Clinical Superiority: Dual agonists outperform GLP-1 monotherapies in weight loss and glycemic control
-
Government and Payer Support: Reimbursement expansion and public health initiatives boost adoption
-
Strategic Collaborations: Partnerships between pharma giants and biotech firms accelerate innovation
-
Patient Preference for Non-Surgical Options: Rising demand for pharmacological alternatives to bariatric surgery